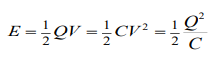ELECTRICITY
The topics covered are:
- monitoring and measuring AC
- current, potential difference, power, and resistance
- electrical sources and internal resistance
- capacitors
- semiconductors and p-n junctions
SUB UNIT HEADING |
DETAILS |
|
Semiconductors and p-n junctions
|
- Knowledge of the terms conduction band and valence band.
- Knowledge that solids can be categorised into conductors, semiconductors or insulators by their band structure and their ability to conduct electricity. Every solid has its own characteristic energy band structure. For a solid to be conductive, both free electrons and accessible empty states must be available. - Qualitative explanation of the electrical properties of conductors, insulators and semiconductors using the electron population of the conduction and valence bands and the energy difference between the conduction and valence bands. (Reference to Fermi levels is not required.) - The electrons in atoms are contained in energy levels. When the atoms come together to form solids, the electrons then become contained in energy bands separated by gaps. - For metals we have the situation where one or more bands are partially filled. Some metals have free electrons and partially filled valence bands, therefore they are highly conductive. Some metals have overlapping valence and conduction bands. Each band is partially filled and therefore they are conductive. - In an insulator, the highest occupied band (called the valence band) is full. The first unfilled band above the valence band is the conduction band. For an insulator, the gap between the valence band and the conduction band is large and at room temperature there is not enough energy available to move electrons from the valence band into the conduction band where they would be able to contribute to conduction. There is no electrical conduction in an insulator. - In a semiconductor, the gap between the valence band and conduction band is smaller and at room temperature there is sufficient energy available to move some electrons from the valence band into the conduction band allowing some conduction to take place. An increase in temperature increases the conductivity of a semiconductor. - Knowledge that, during manufacture, semiconductors may be doped with specific impurities to increase their conductivity, resulting in two types of semiconductor: p-type and n-type. - Knowledge that, when a semiconductor contains the two types of doping (p-type and ntype) in adjacent layers, a p-n junction is formed. There is an electric field in the p-n junction. The electrical properties of this p-n junction are used in a number of devices. - Knowledge of the terms forward bias and reverse bias. Forward bias reduces the electric field; reverse bias increases the electric field in the p-n junction. - Knowledge that LEDs are forward biased p-n junction diodes that emit photons. The forward bias potential difference across the junction causes electrons to move from the conduction band of the n-type semiconductor towards the conduction band of the ptype semiconductor. Photons are emitted when electrons ‘fall’ from the conduction band into the valence band either side of the junction. - Knowledge that solar cells are p-n junctions designed so that a potential difference is produced when photons are absorbed. (This is known as the photovoltaic effect.) The absorption of photons provides energy to ‘raise’ electrons from the valence band of the semiconductor to the conduction band. The p-n junction causes the electrons in the conduction band to move towards the n-type semiconductor and a potential difference is produced across the solar cell. Vertical Divider
|
Summary Notes & Questions
Remember to continual check your past paper attempts and review unknown areas with your summary notes. Websites like BBC Bitesize and Mr MacKenzie Fizzics will also help your revision. Ask your teacher if you are unsure of how to proceed with your revision plan and to clarify areas you are unsure about.